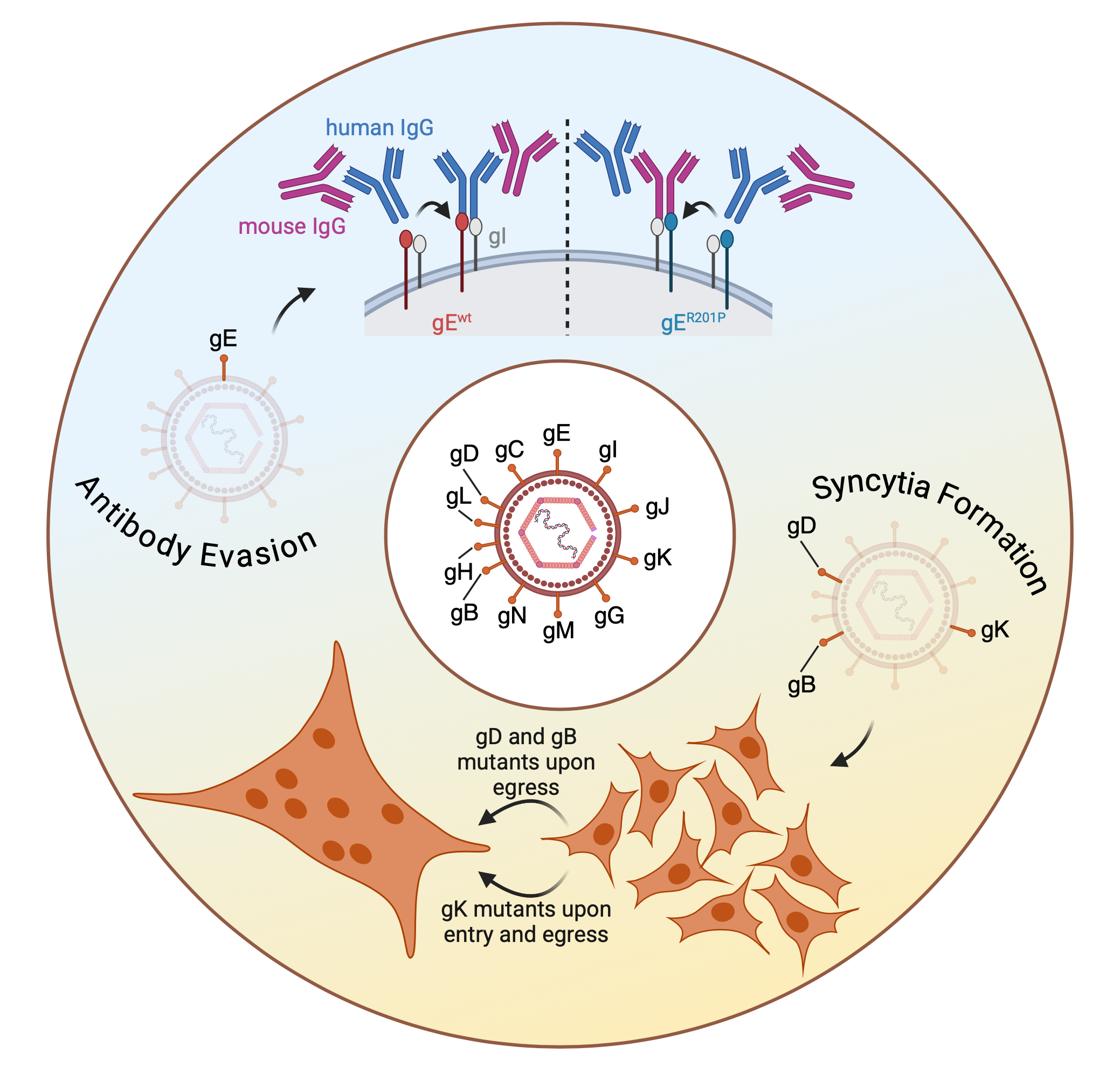
Glycoproteins of enveloped viruses are their outer most layer, and therefore their means to interact with cells and surfaces. This study highlights the evolutionary plasticity of this group of proteins, but also how they facilitate social interactions between co-infecting viruses.
Syncytial Plaques
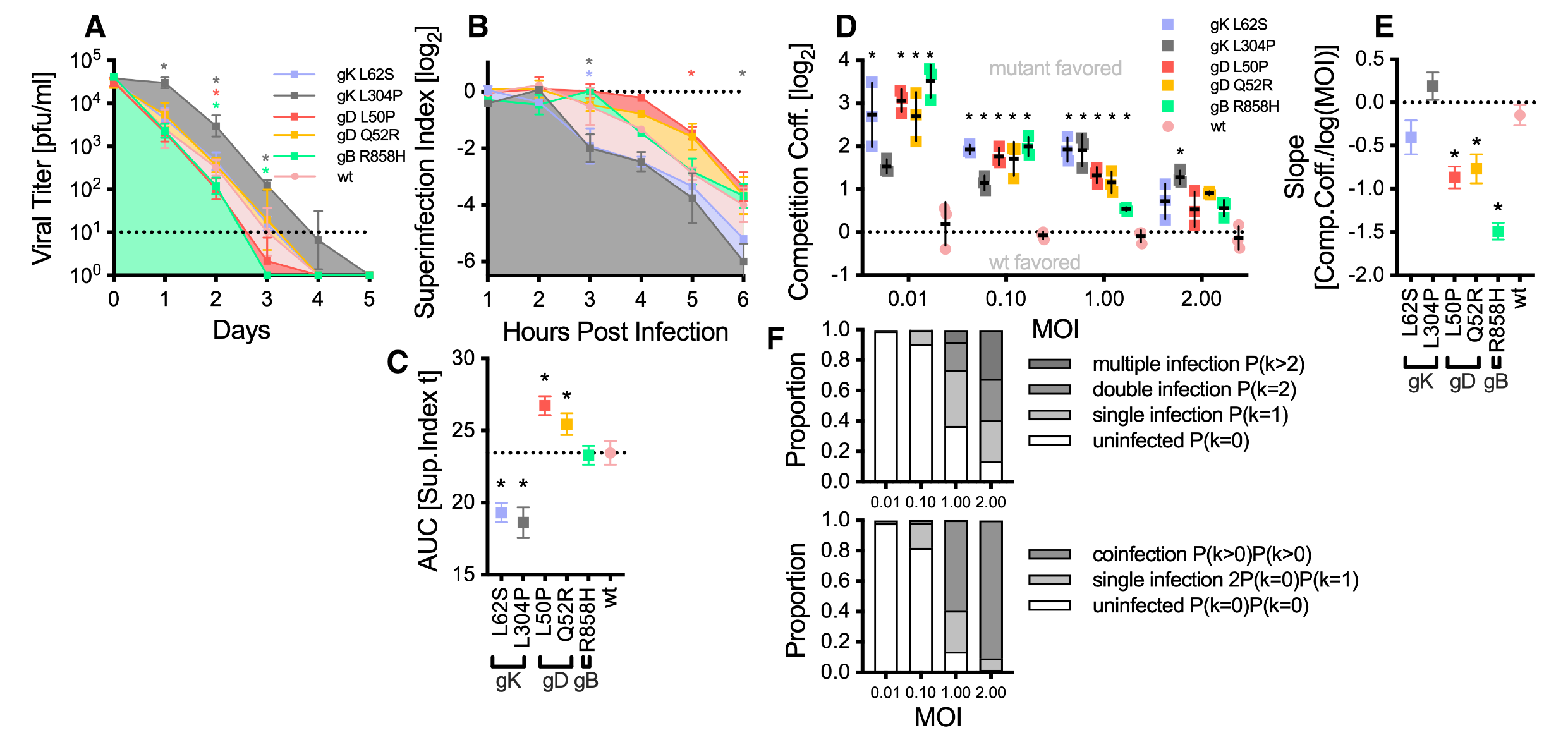
Using the same hypermutator virus as previously published (https://doi.org/10.1093/molbev/msae119) we could select for viral population with syncytial plaque phenotypes, but also antibody resistant viruses. Mutations in glycoproteins (g)D, gK and gB do facilitate syncytia formation, but they also influence particle stability and superinfection exclusion.
Anti-gD Antibody Resistance is a Social Phenotype
Resistance to anti-gD antibodies, however, was not mediated by gD, but by gE mutations. The gI/E complex is a known Fc receptor important for bipolar bridging of antibodies, a mechanism to clear them effectively. Our observed gE mutants enabled recognition of mouse Fc, allowing for antibody evasion without changing the receptor binding domain. Strikingly, this occurs in a social manner, as co-infection of both genotypes (wt and gE) rescues wt virus.
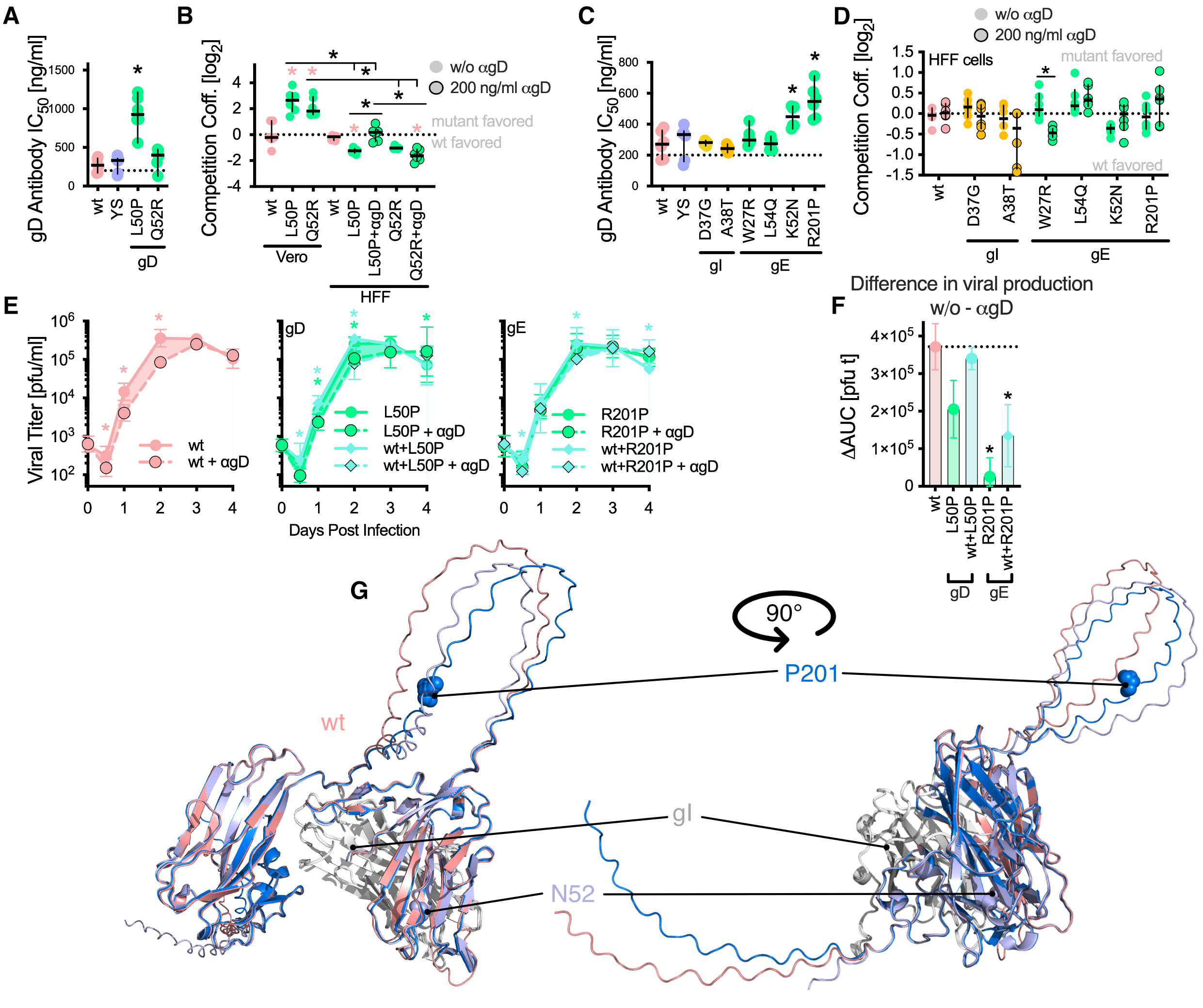
Glycoproteins do diversify rapidly. They are key players in cell culture adaptation, modify cell fusion and evade antibodies. Additionally, glycoprotein mutants also enable social interaction within viral populations, both beneficial (antibody evasion) and antagonistic (superinfection exclusion). For further information please see the original publication https://doi.org/10.1093/ve/veaf072.